Free-Radical Polymerization of Allyl Monomers
Allyl monomers are compounds with the structural formula H2C=CH−CH2R, where R is the rest of the molecule. Unlike vinyl monomers, these compounds polymerize only slowly and yield only low molecular weight polymers or oligomers.1-3
Allyl polymerization has received much less attention than conventional vinyl polymerization. Most of the published articles focus on the polymerization and copolymerization of allyl compounds by conventional methods such as free radical polymerization and photopolymerization. It was found that allyl radicals are much more stable than corresponding vinyl radicals and that propagation is quickly terminated by hydrogen abstraction from an allylic monomer:

The self-termination by allylic hydrogen atom abstraction is often referred to as allylic degradative chain transfer. The alylic monomer radical stabilizes itself by resonance which explains the slow polymerization of allylic monomers and the relative low molecular weight of the polymers.

Similar difficulties were observed when copolymerizing allylic monomers with vinylic monomers. Consequently, to polymerize allylic monomers large quantities of free radical initiators are required, resulting in relatively large amounts of terminal initiator end groups and residual initiator decomposition products.
| Compound | Repeating Unit | Boiling Point (°C) |
| Allyl Acetate |
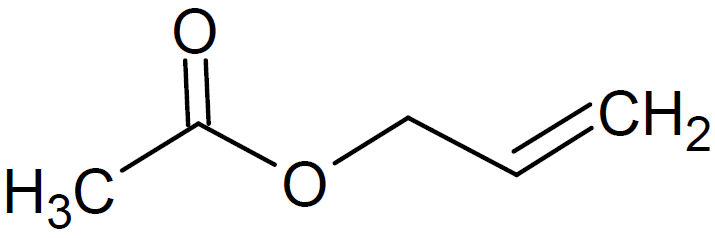 |
195 |
| Allyl Alcohol |
|
111 |
| Allyl benzyl ether |
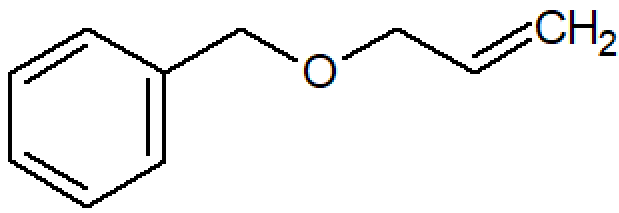 |
457 |
| Allyl Phenyl Ether |
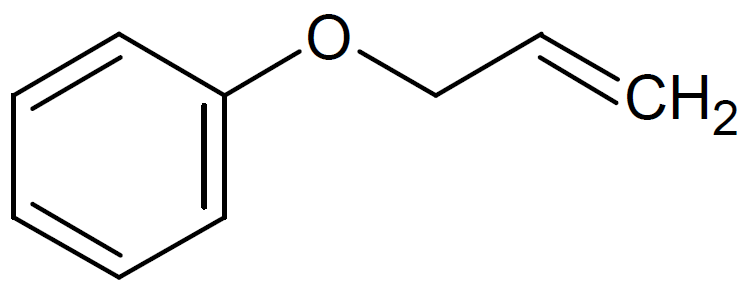 |
188 |
| 3-Allyloxy-1,2-propanediol |
|
N/A |
Notes & References
P.D. Bartlett, and F. A. Tate, J. Am. Chem. Soc. 75 (1), pp 91-95 (1953)
P.D. Bartlett, and R. Altschul, J. Am. Chem. Soc. 67 (5), pp 812-816 (1945)
V.I. Volodina, A.I. Tarasov and S.S. Spasskii, Poly. Sci. U.S.S.R., 18 (10), pp 2658-2662 (1976)
M. Litt and F.R. Eirich, J. Poly. Sci. 45, pp 379-396 (1960)
A. Matsumoto, T. Kumagai, H. Aota, H. Kawasaki, and R. Arakawa, Poly. J., 41, 26-33 (2009)
- Sigma Allyl Monomers